The case n = 1 corresponds to an isothermal (constant-temperature) process for an ideal gas and a polytropic process. .png)
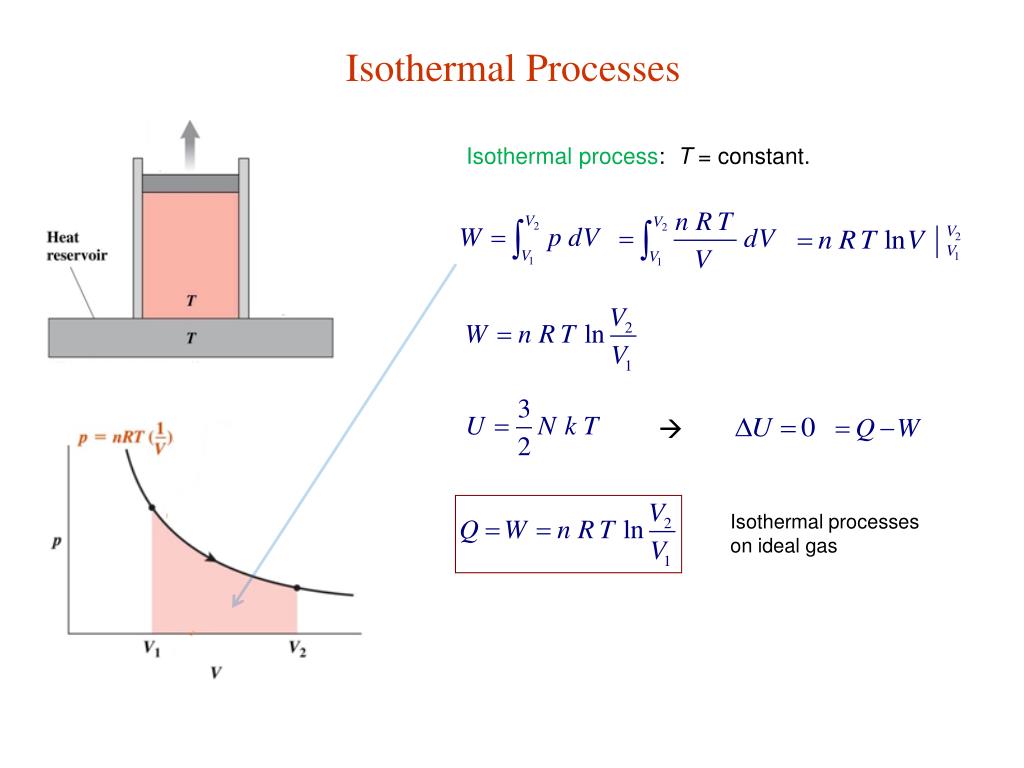
In each of these states, the thermal equilibrium is maintained. The heat transfer into or out of the system typically must happen at such a slow rate to continually adjust to the temperature of the reservoir through heat exchange. Since entropy is a state function (that depends on an equilibrium state, not depending on a path that the system takes to reach that state), the change in entropy of the system is the same as in the reversible process and is given by the formulas above.An isothermal process is a thermodynamic process in which the system’s temperature remains constant (T = const). Such an expansion is also isothermal and may have the same initial and final states as in the reversible expansion. Once obtained, these formulas can be applied to an irreversible process, such as the free expansion of an ideal gas. In thermodynamics, the reversible work involved when a gas changes from state A to state B is W A → B = − ∫ V A V B p d V. The purple area represents the work for this isothermal change. Thus, specifying that a process is isothermal is not sufficient to specify a unique process.įigure 2. For an ideal gas, this means that the process is also isothermal. a free expansion, there is no change in internal energy. For details of the calculations, see calculation of work.įor an adiabatic process, in which no heat flows into or out of the gas because its container is well insulated, Q = 0. In either case, with the aid of a suitable linkage the change in gas volume can perform useful mechanical work. For isothermal expansion, the energy supplied to the system does work on the surroundings. If the gas is ideal, the amount of energy entering the environment is equal to the work done on the gas, because internal energy does not change. To maintain the constant temperature energy must leave the system as heat and enter the environment. Doing work on the gas increases the internal energy and will tend to increase the temperature. In the isothermal compression of a gas there is work done on the system to decrease the volume and increase the pressure. Note that this is true only for ideal gases the internal energy depends on pressure as well as on temperature for liquids, solids, and real gases. This is a result of the fact that in an ideal gas there are no intermolecular forces. Thus, in an isothermal process the internal energy of an ideal gas is constant. This is a consequence of Joule's second law which states that the internal energy of a fixed amount of an ideal gas depends only on its temperature. Isothermal processes are of special interest for ideal gases. Isothermal processes are often used and a starting point in analyzing more complex, non-isothermal processes. Phase changes, such as melting or evaporation, are also isothermal processes when, as is usually the case, they occur at constant pressure. In the thermodynamic analysis of chemical reactions, it is usual to first analyze what happens under isothermal conditions and then consider the effect of temperature. Some parts of the cycles of some heat engines are carried out isothermally (for example, in the Carnot cycle). Isothermal processes can occur in any kind of system that has some means of regulating the temperature, including highly structured machines, and even living cells. The adjective "isothermal" is derived from the Greek words "ἴσος" ("isos") meaning "equal" and "θέρμη" ("therme") meaning "heat".
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
